
To reduce their instability, most of the developed siRNA agents carry chemical modifications in their nucleotide sugars, nucleobases, or phosphodiester linkages to prevent RNase cleavage 5, 6, 7. Although siRNA-based therapeutics have enormous potential for silencing specific genes that cannot be targeted by existing drugs, the development of siRNA-based drugs has been faced several difficulties: prevention of rapid degradation by nucleases, delivery to the target tissue, entry into the target cells, avoidance of innate immune responses and reduction of off-target effects 3, 4. RNA interference (RNAi) is a gene regulatory mechanism in which the expression of specific genes is silenced by endogenous double-stranded RNAs or by synthetic short interfering RNAs (siRNAs) consisting of oligoribonucleotide duplexes of 21–23 bases 1, 2, 3. Collectively, these findings demonstrate that ODAGal4 is a potent stabiliser of siRNAs, particularly nucleotides with phosphorothioate linkages, representing a promising tool in the development of gene-silencing therapies. 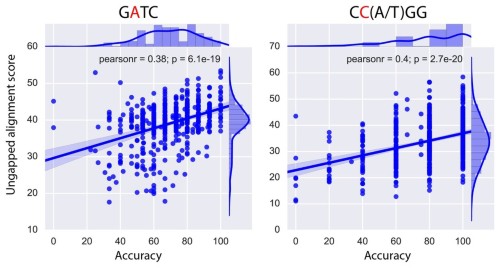
Importantly, ODAGal4 did not interrupt gene-silencing activity of all the RNAs tested. Furthermore, melting temperature analysis showed that ODAGal4 greatly increases the thermal stability of phosphorothioate RNAs. Serum degradation assays of RNAs with multiple chemical modifications showed that ODAGal4 preferentially improves the stability of RNAs with phosphorothioate modification among chemical modifications. The half-lives of fully phosphorothioate-modified RNA duplexes with ODAGal4 were more than 15 times longer than those of unmodified siRNAs without ODAGal4 this improvement in serum stability was superior to that observed for other chemical modifications. In particular, a combination of ODAGal4 and incorporation of phosphorothioate linkages into the siRNA prominently prevented degradation by serum. The effect of ODAGal4 on siRNA stabilisation was further amplified by introduction of modified nucleotides into the siRNA. ODAGal4 improved the resistance of various siRNAs against serum degradation. To improve siRNA stability, we synthesised a double-stranded RNA-binding cationic oligodiaminogalactose 4mer (ODAGal4) and investigated here its characteristics for siRNA stabilisation in vitro. Collectively, our findings demonstrate a functional cooperation between USP8, AIP4, and the ESCRT-0 machinery at the early sorting phase of CXCR4 and underscore the versatility of USP8 in shaping trafficking events at the early-to-late endosome transition.Small interfering RNAs (siRNAs) are potential tools for gene-silencing therapy, but their instability is one of the obstacles in the development of siRNA-based drugs.  
USP8 functionally opposes the ubiquitin ligase AIP4 with respect to ESCRT-0 ubiquitination, thereby promoting trafficking of CXCR4. Perturbation in CXCR4 trafficking, resulting from USP8 inactivation, occurs at the ESCRT-0 checkpoint, and catalytic mutation of USP8 specifically targeted to the ESCRT-0 complex impairs the spatial and temporal organization of the sorting endosome. In the presence of ligand, diminished CXCR4 turnover is accompanied by receptor accumulation on enlarged early endosomes and leads to enhancement of phospho-ERK signaling. Contrary to the effects of USP8 loss on enhanced green fluorescent protein, we find that USP8 depletion stabilizes CXCR4 on the cell surface and attenuates receptor degradation without affecting its ubiquitination status. The present study explores an indirect role for USP8 in cargo trafficking through its regulation of the chemokine receptor 4 (CXCR4). Ubiquitin-specific protease 8 (USP8) has previously been implicated in endocytosis of several receptors by virtue of their deubiquitination. Reversible ubiquitination orchestrated by the opposition of ubiquitin ligases and deubiquitinating enzymes mediates endocytic trafficking of cell surface receptors for lysosomal degradation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
